Molecules of the same type follow this trend. While the molecular weight is the sum of the atomic weights of the elements present in the molecule.įor elements, the greater the molar mass, the greater the boiling point. The molecular mass is the sum of the atomic masses of the elements present in the molecule. It is calculated by adding the number of protons and neutrons (electrons are considered too light and are negligible). The atomic mass is the mass of a single atom/isotope. 1 dozen 12 items similarly we use the mole to. Just as we take a standard value to calculate different things e.g. 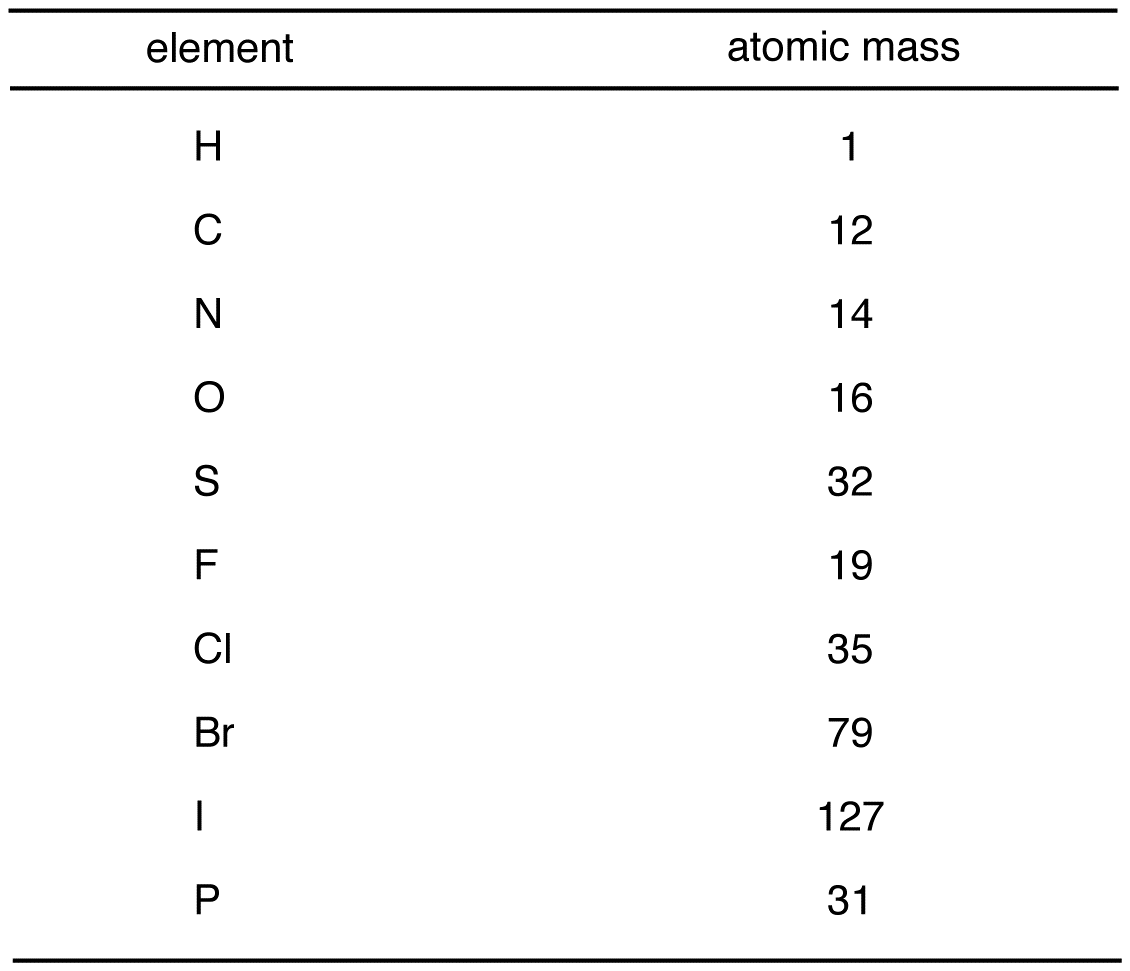
A mole of any substance is 6.022×1023 molecules. atoms, molecules, ions, is defined as a mole. In a substance, the amount of entities present e.g. The atomic weight of an element is an average of the weight of its isotopes with respect to their relative abundances. Molar mass of a substance is the mass in grams of one mole of the compound. The molecular mass would then be: $$(2*1.01\frac$$ Meyer aligned the elements in his table according to periodic variations in simple atomic properties, such as atomic volume (Figure 7. Water is made up of 2 hydrogens and 1 oxygen, and the atomic mass of hydrogen is 1.01 g/mol and the atomic mass of oxygen is 16.00 g/mol. The molecular weight of water can be calculated by adding together the atomic masses of the elements in it. Thus, the context and questions must be clear when discussing these terms! Always be careful when reading textbooks and problems to see if when "molecular mass" is mentioned they actually mean "molecular mass" or if they mean "molecular weight" instead.Īs a guide to get you used to this, if I am using a term with its original definition, it will be in green, if I am using it with its colloquial definition it will be in red. The Mole How are chemists able to measure things when everything is made up of different atoms and, to make this more difficult, different isotopes of those atoms? With moles, of course! Not with the small subterranean mammal (though they are cute), but the unit moles.Īs I mentioned before, atomic mass and atomic weight are often used interchangeably, so molar mass, molecular mass, AND molecular weight are also often used interchangeably. Lastly, we will look at the link between molar mass and boiling point.Then, we will explain the concept of molecular mass and explain the differences between it and molar mass.Next, we will explain the concepts of atomic mass and molar mass and the differences between the two.First, we will define a mole and explain why that unit is used.This article is about moles and molar mass.In this article, we will discuss how chemists measure and group the masses of atoms and molecules. However, we first need to know the average mass of each atom. We treat atoms the same way! To find out how many atoms are in a sample, we can relate the mass of the sample to the number of atoms. If you know the mass, you can multiply that by the desired quantity and add the candy to a scale until you reach that weight. This is a very long and tedious way to do business! Instead, you utilize the average mass of the candy. Metals are by far the largest of the three classes.Let's say you work at a candy store and a customer asks for different amounts of candy (100, 250, 1000…), so you count out each order. Normally, however, molar volume is expressed in units of cm 3. They are one of three classes of elements (the other two classes are nonmetals and metalloids). The molar volume is also known as the atomic volume. 6.5: Metals Metals are elements that can conduct electricity.6.4: Modern Periodic Table- Periods and Groups.For example, the atomic mass of copper is 63.546 amu. This is why most of the atomic masses on the periodic table are not exact numbers. When we compare the noble gas relative atomic masses M r, we get: M r,He 4.0026. The atomic masses indicated on the periodic table represents an average mass for each element based on the proportion of each isotope present on the Earth. Since this occurs as you go down a group, the atomic mass increases as you go down a group. Both can be used to solve problems that would be hard to do by eye. So, when the atomic number increases, the mass number of the isotopes is higher (though they are different isotopes), and so the relative atomic mass will increase. The first conversion factor can be used to convert from mass to moles, and the second converts from moles to mass. 6.1: Early History of the Periodic Table 1 molAl 26.98g Al and 26.98gAl 1mol Al (5.4.1) (5.4.1) 1 m o l A l 26.98 g A l a n d 26.98 g A l 1 m o l A l. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
